Innovative Cancer Treatment May Sometimes Cause Cancer, F.D.A. Says
The News
A lifesaving cancer treatment may itself cause cancers, the Food and Drug Administration reported on Tuesday.
The treatment, called CAR-T was first approved in November 2017 for life-threatening blood cancers. But, the F.D.A. said, it had received 19 reports of new blood cancers in patients who received the treatment.

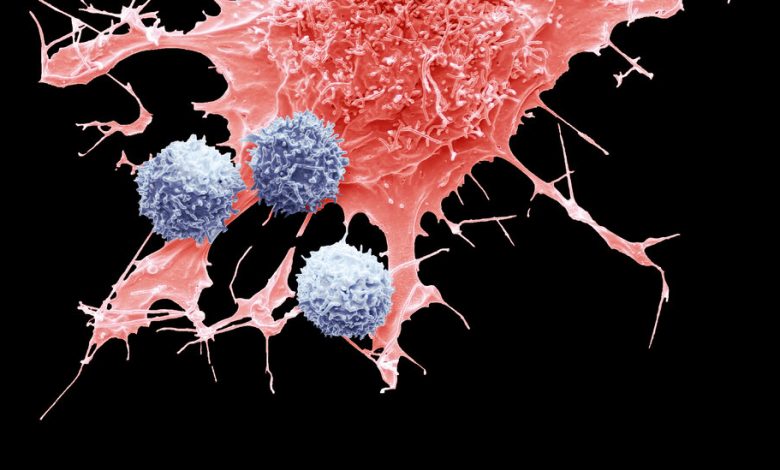
A composite colored scanning electron micrograph of T cells, blue, and a lymphoma cancer cell, red.Credit…Steve Gschmeissner/Science Source
A Number That Sums It Up: Thousands of lives have been saved with CAR-T.
CAR-T involves removing a type of white blood cell — T cells — from a patient’s blood, then genetically engineering to make proteins — chimeric antigen receptors (CAR) — which allow the T cells to attach to cancer cells and kill them. The engineered cells are then infused back into the patient’s blood.
The F.D.A. has approved six commercial CAR-T products. Cancer specialists said the treatments have saved the lives of thousands of patients with blood cancers. Even if there is a causal link between the treatments and a small risk of a new blood cancer, the regulators said on Tuesday, the benefits of the treatment outweigh the risks. That sentiment was echoed by doctors involved in cancer treatment.
While the hypothetical risk, was known, “we haven’t observed it” in patients, Dr. Marcela V. Maus, director of cellular immunotherapy at Massachusetts General Hospital, said.
Dr. John DiPersio, director of the center for genetic and cellular immunotherapy at Washington University School of Medicine in St. Louis, said his center had treated 500 to 700 patients. And, he said, “I haven’t seen a single one” develop a new T cell cancer.
CAR-T therapy has been reserved for patients who would die without it, he added.
“They are all going to die and they are all going to die quickly without this treatment. It saves their life,” Dr. DiPersio said. “It works in a substantial portion of patients. The benefit is enormous.”
Facts to Keep in Mind: What triggered the F.D.A.’s investigation.
The F.D.A. said in its announcement that the reports of additional cancers included serious consequences — hospitalizations and deaths. And, the agency said, it is known that the way CAR-T cells are produced has a risk of causing cancers in recipients.
When patients’ T cells are engineered to make proteins that attack cancer cells, a virus helps slip new genes into T cell DNA. That has the potential to disrupt other genes, leading to cancer.
But there are other potential explanations. CAR-T therapy is used when patients have already had at least one round of conventional treatments with intense chemotherapy and, often, radiation. Those treatments can themselves elicit new blood cancers. Even without chemotherapy or radiation, Dr. Maus added, patients with blood cell cancers are especially susceptible to developing other blood cell cancers.
What Happens Next: The search for a smoking gun.
One unanswered question, Dr. Maus and Dr. DiPersio said, is whether the new cancers involved T cells carrying the added CAR proteins. That does not prove the gene insertion caused the cancers. But Dr. DiPersio said, “it is more of a smoking gun.”
The F.D.A. did not describe any anticipated outcomes of its investigation but said that it was “evaluating the need for regulatory action.”




